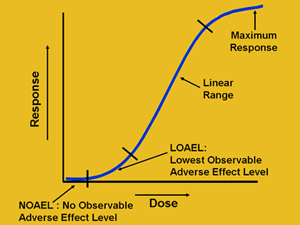
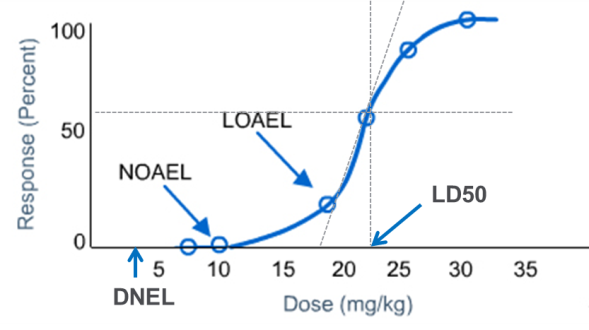
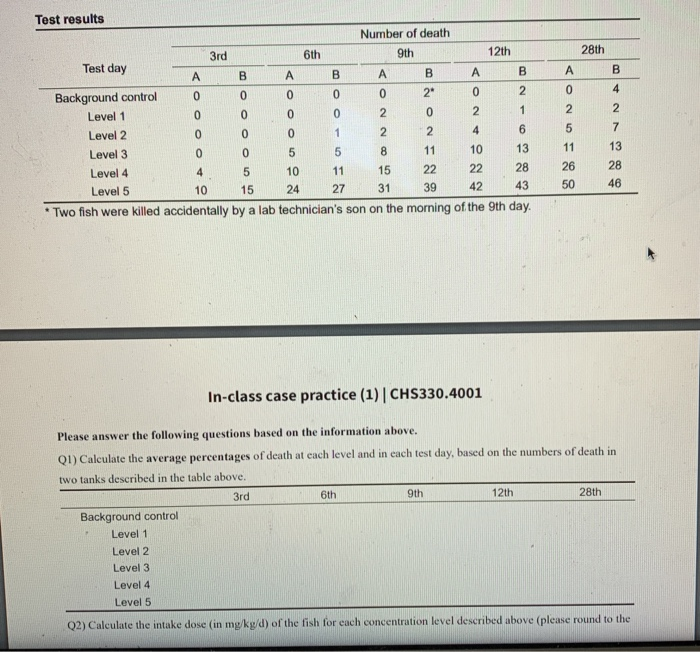
A PROPOSAL FOR CALCULATING THE NO-OBSERVED-ADVERSE-EFFECT LEVEL (NOAEL) FOR ORGANIC COMPOUNDS RESPONSIBLE FOR LIVER TOXICITY BA
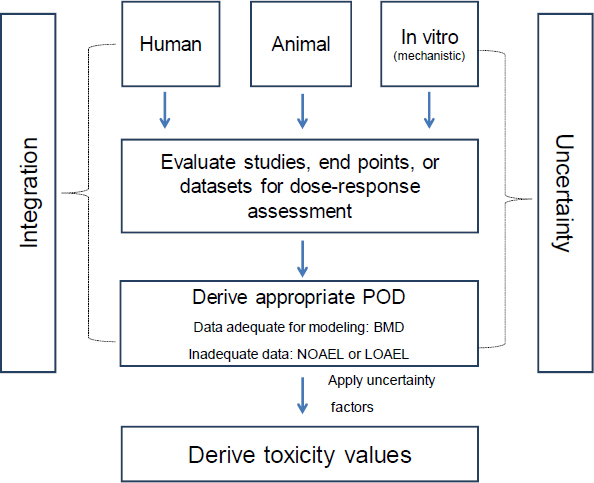
7 Derivation of Toxicity Values | Review of EPA's Integrated Risk Information System (IRIS) Process |The National Academies Press

Calculation of the FIH dose from MABEL and NOAEL. The FIH dose from... | Download Scientific Diagram

Hormesis Shifts the No-Observed-Adverse-Effect Level (NOAEL) - Evgenios Agathokleous, Costas Saitanis, Athina Markouizou, 2021

Determination of starting dose of the T cell-redirecting bispecific antibody ERY974 targeting glypican-3 in first-in-human clinical trial | Scientific Reports

Practical Considerations in Determining Adversity and the No-Observed-Adverse-Effect-Level (NOAEL) in Nonclinical Safety Studies: Challenges, Perspectives and Case Studies - Vijay P. Kale, Ilona Bebenek, Hanan Ghantous, John Kapeghian, Bhanu P. Singh ...

Calculation of the FIH dose from MABEL and NOAEL. The FIH dose from... | Download Scientific Diagram
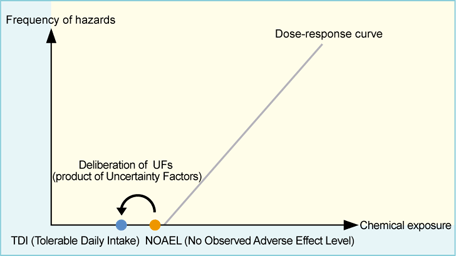
Risk Assessment on chemicals-For Better Understanding-4 | Chemical Management | National Institute of Technology and Evaluation (NITE)







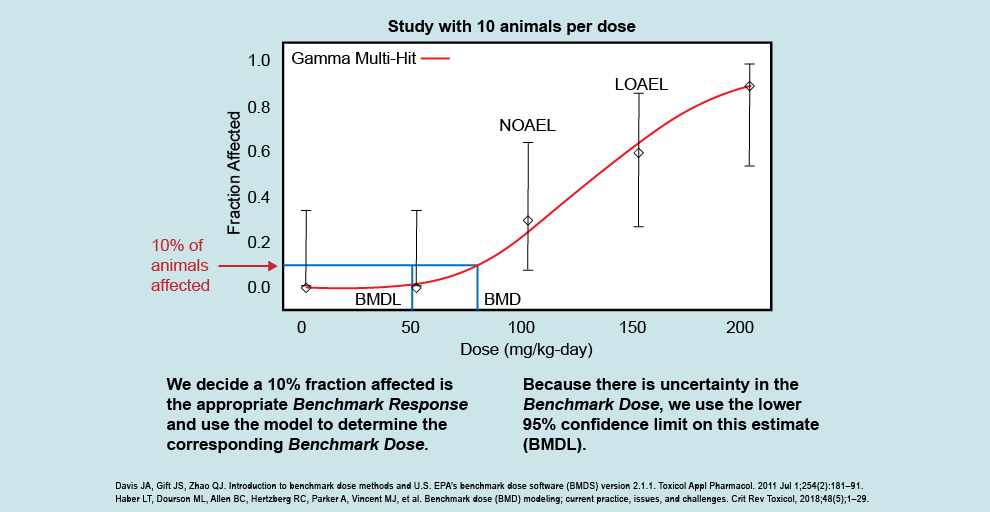



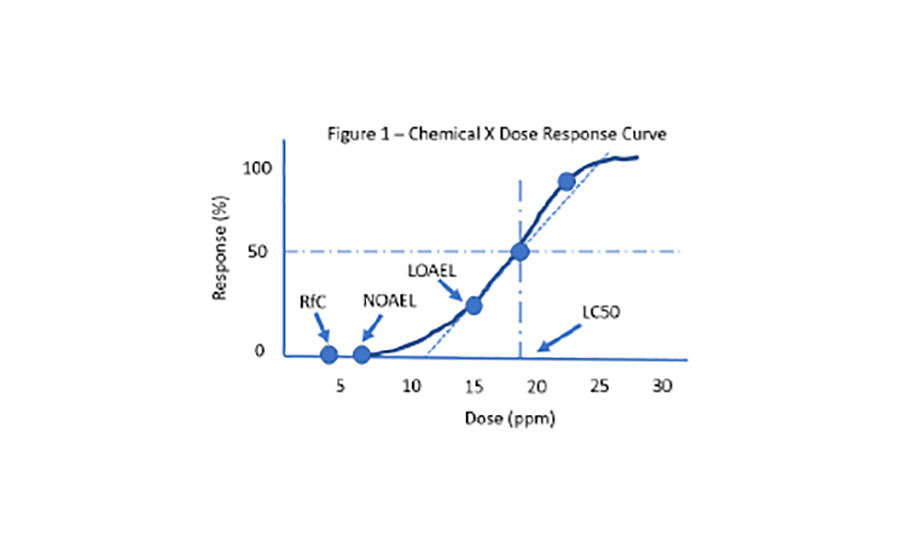

.png)